Basic QC Practices
Latin and South American QC Survey Results
In 2017, we conducted a Spanish language QC Survey in the 16 countries in Latin and South America. What do their results tell us about the state of quality control? Are they doing better QC than the US?
The Great Global QC Survey: Results from Latin and South America
November 2017
Sten Westgard, MS
For a free copy of these graphs, go to our download section.
After looking at the breakdown of the Great Global QC Survey results between US and outside the US, we decided to go one step further. Knowing that many countries in Latin and South America are primarily Spanish-speakers, with less English in their vocabulary, we built a purely Spanish-language QC Survey. Over the course of a few months, we gathered 100 responses from 16 different countries.

Unlike the US, where mostly private hospital laboratories responded to the survey, the most common type of labs responding to this survey were public laboratories. That makes sense when we consider that many healthcare systems in the region are socialized and run as public institutions.
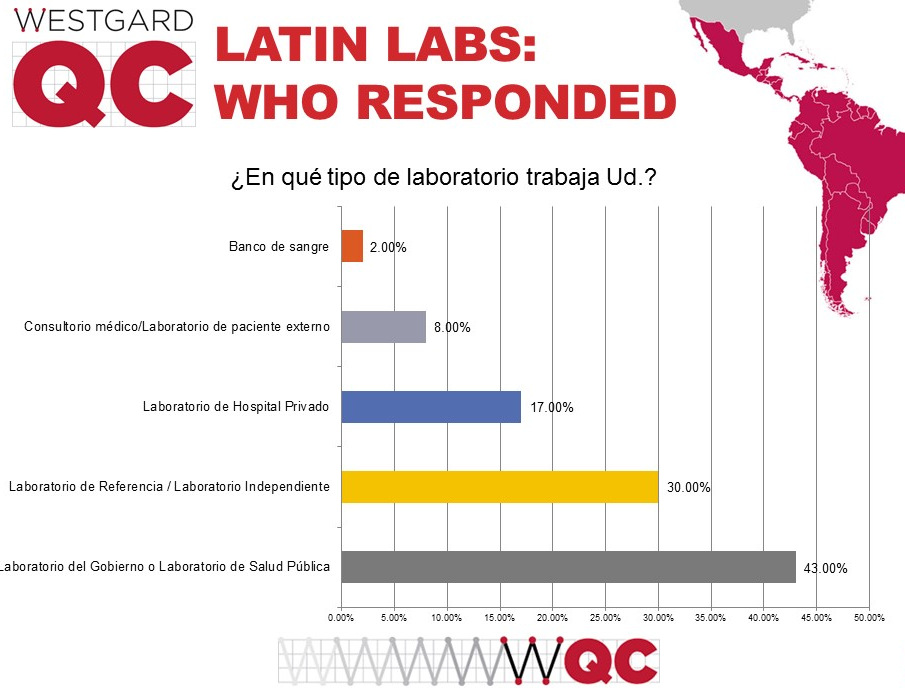
Now, what was the volume of testing in these responding laboratories?
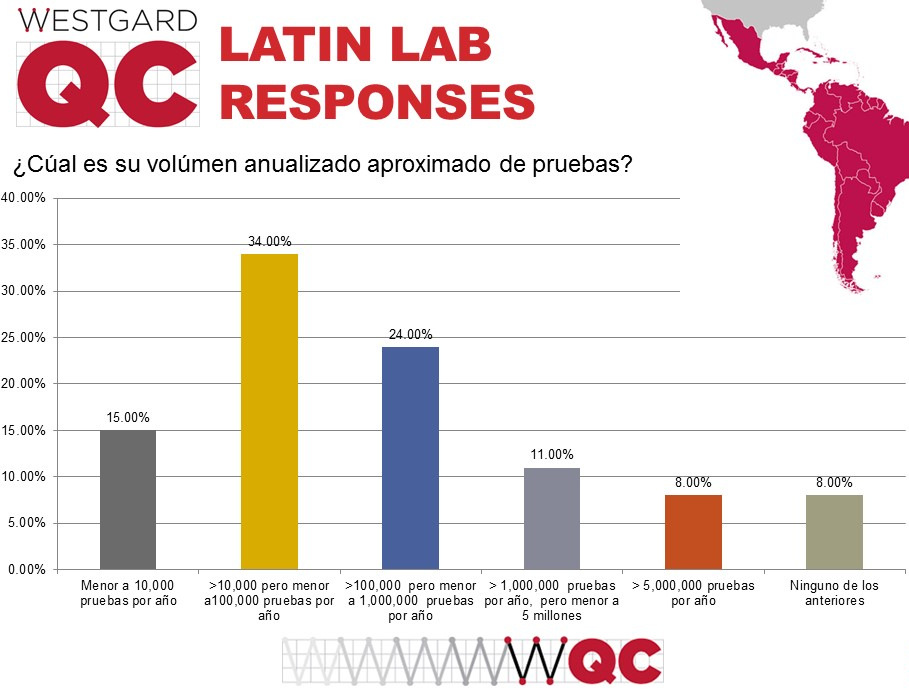
The breakdown of the laboratories is unlike the US and global distribution of responses. There is a longer "tail" out toward the higher volumes, while the most common lab volume is between 10,000 and 100,000 tests, an order of magnitude smaller than the most common volume in our other surveys. So we have smaller labs responding, with a few giant "outlier" laboratories.
Let'sget right to the QC practices of these laboratories:
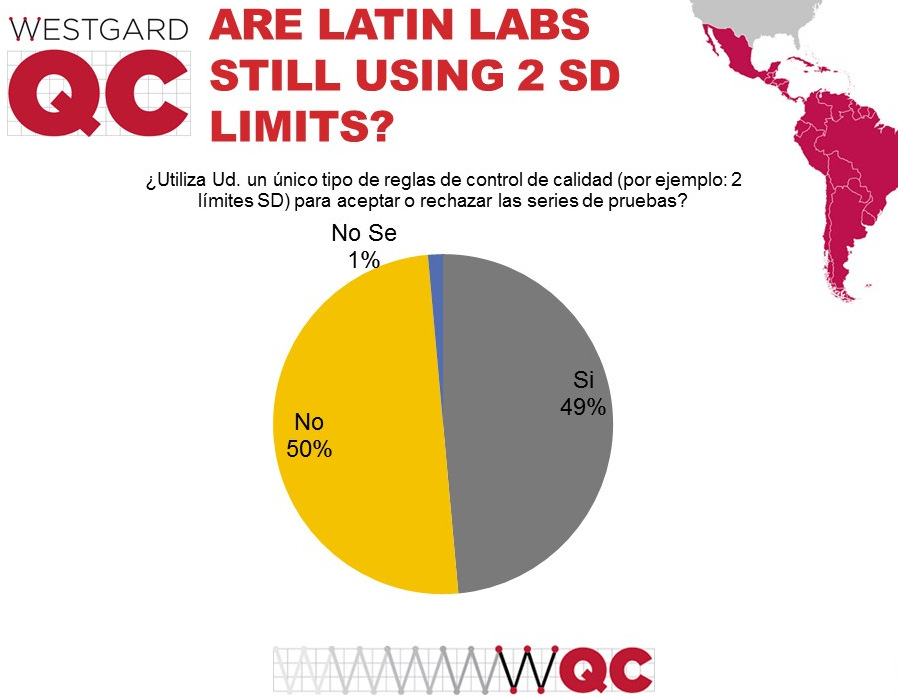
A slight difference here from the other surveys: fewer labs in Latin and South America are using the old traditional 2 SD control limits. While a majority of labs in the US and globally use the antiquated approach, these results show just under half of labs are using 2 SD limits.
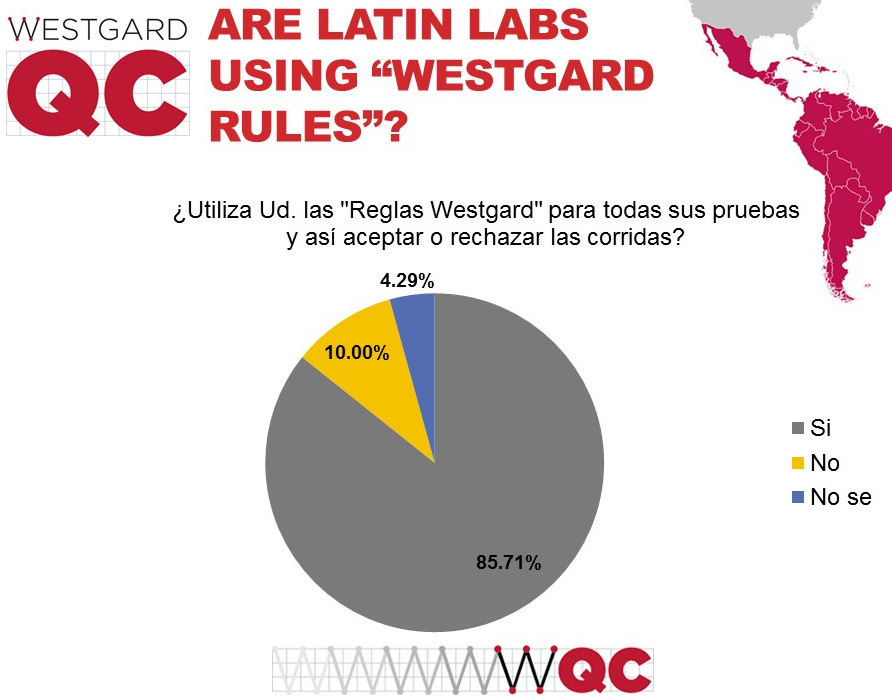
A much higher percentage of labs are using "Westgard Rules" in Latin and South America than in the US or globally. The use in the over global survey was around 75%. We suspect that the stronger professionalism of laboratorians in Latin and South America motivates them to adopt more sophisticated approaches to quality control. There may be cases where labs are using too many "Westgard Rules" but that will depend on their analytical performance.
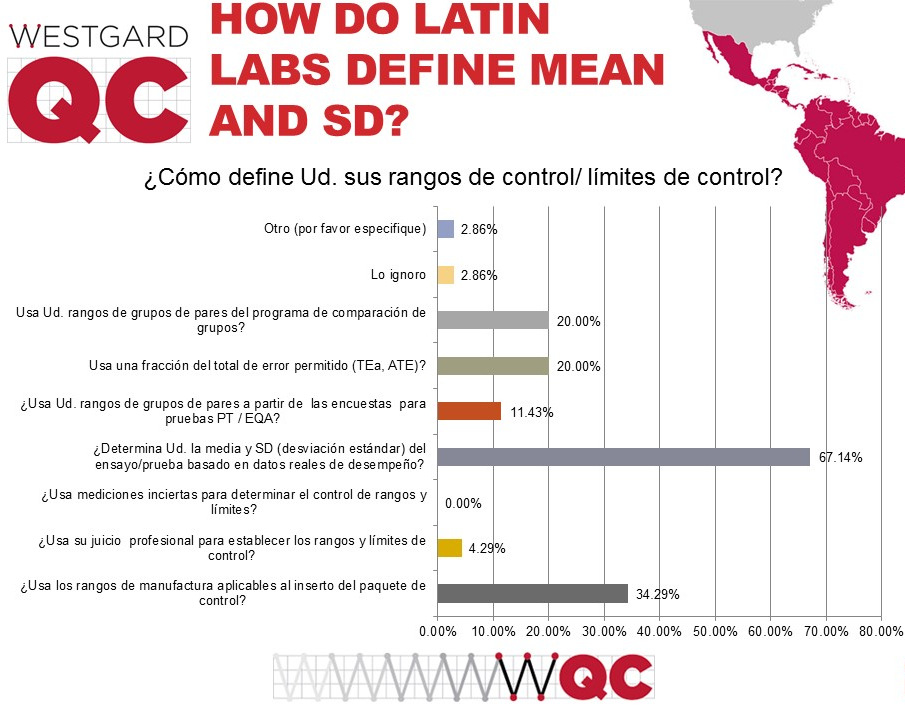
Two-thirds of laboratories actually use their own mean and standard deviation when setting up their control limits, ranges, and charts. This is encouraging. Nevertheless, you can see a third of laboratories will also employ manufacturer ranges on their testing. Those manufacturer ranges are typically too wide, sometimes more than twice as wide as the should be if they reflected the actual SD of the laboratory.
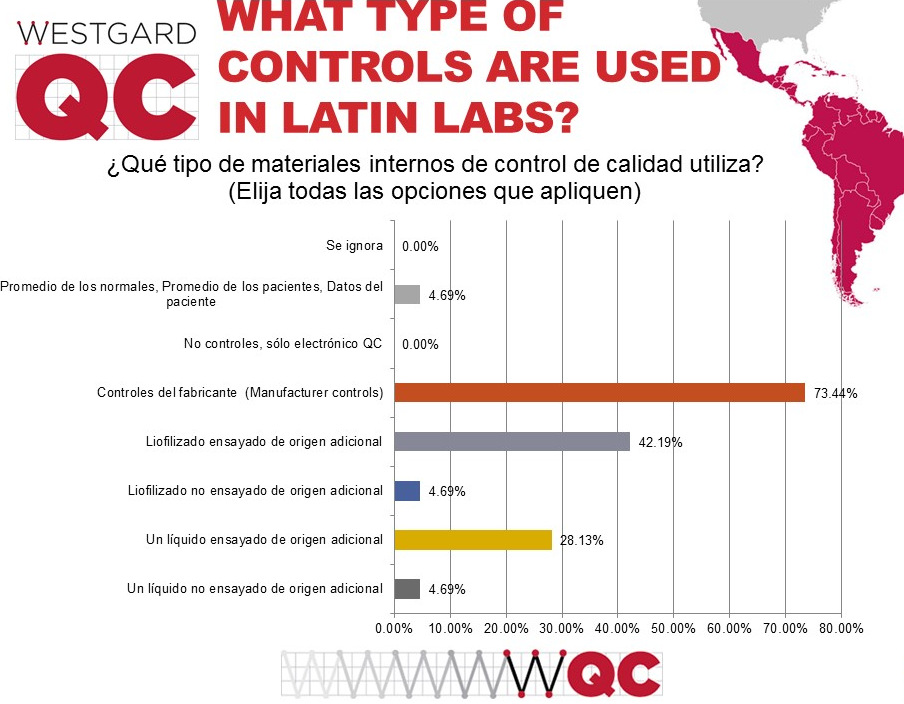
Nearly three-quarters of laboratories are using manufacturer controls, significantly higher than globally, despite the strong recommendation from ISO 15189 and other guidelines to use independent, third-party controls. The second-most popular type of control in use around the world is the lyophilized, assayed control. In the US, liquid controls are more popular than lypophilized, but outside the US the opposite is true. For many labs that have challenging logistical chains, a lyophilized control can be more reliable than a liquid control. Other significant findings here include (1) only a small number of labs are implementing average of normals procedures, despite all the talk and discussion about them over the past decade; and (2) no laboratories are using just electronic QC. In the US, where POC devices are proliferating, the electronic QC is often the only practical method of controlling quality. Outside the US, POC usage is not as high, and so fewer labs use electronic QC.
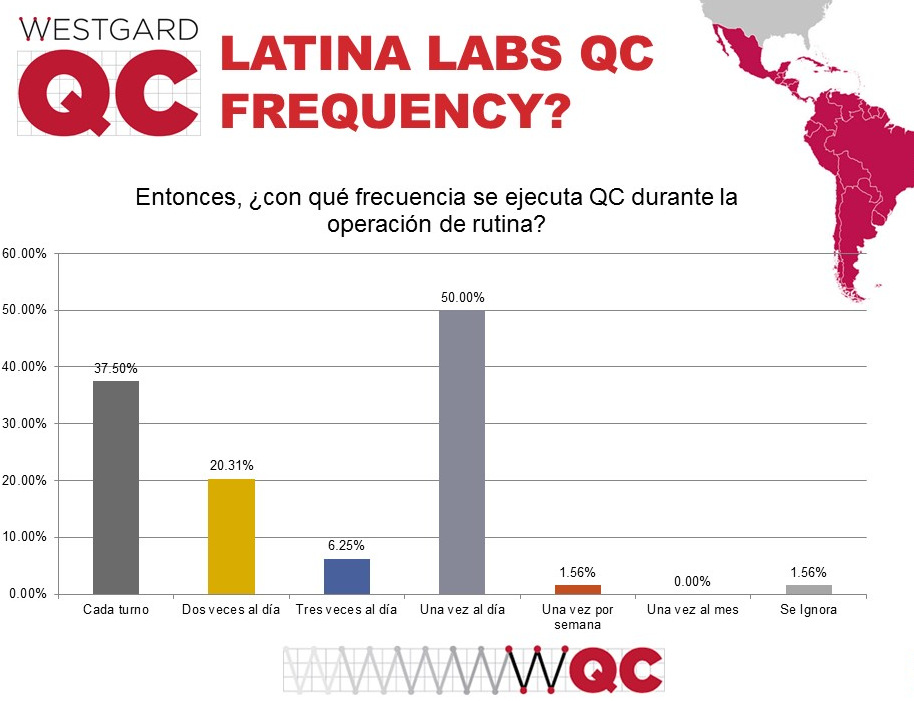
A majority of labs that responded are conforming to the tradition of running QC once a day. There is a significant number of labs that run QC once per shift.
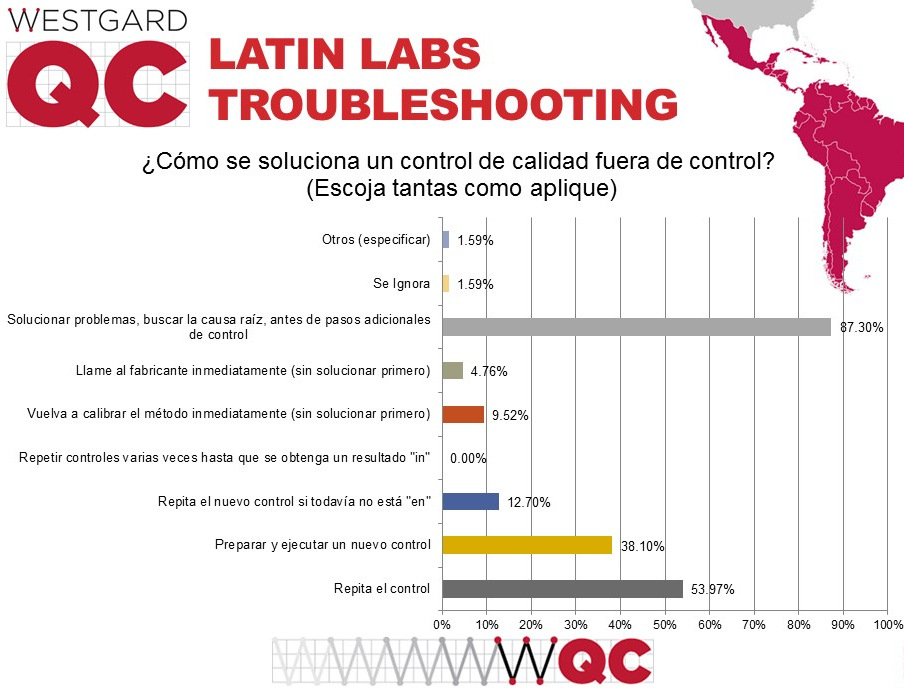
When there is an out-of-control flag, nearly ninety percent of labs in the region respond with the correct action: trouble-shoot first, seek a root cause, etc., before trying any repeated controls. That is a significantly higher percentage than US and global labs. But a majority of labs also still respond to some out-of-control events by merely repeating of the control, following by almost 40% of laboratories willing to run a new control if the repeat is still out, then followed again by 12% of labs repeating the new control even if it's still out.
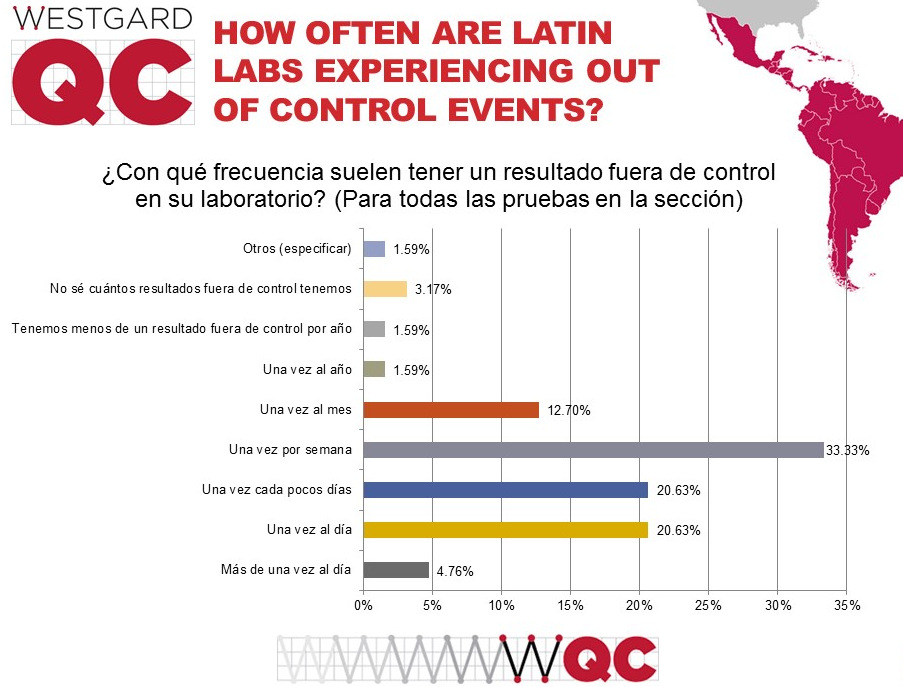
1 in 4 laboratories in Latin and South America report being out -of-control every day, or multiple times per day. That's lower than the global rate of 1 in 3 labs. But while the daily rate is lower, the weekly rate is higher: three out of four laboratories experience multiple flags each week. This is a very high number of out-of-control flags, and that amount of "noise" risks defeaning the laboratory workers. They will suffer from "alert fatigue" and begin ignoring the errors or treating them as less serious.
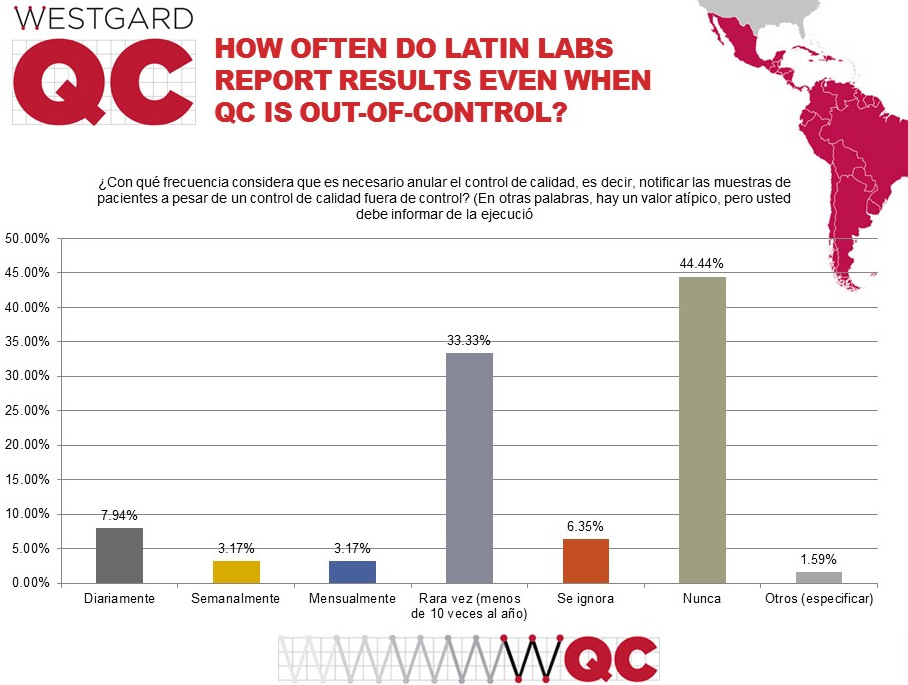
Here's the most critical question: How do all the poor practices, repeats, range errors, etc impact the ultimate purpose of QC? It's not good. Nearly 1 in 10 labs admit to releasing patient results every week (or more frequently), even when they have an out-of-control result. This is the same rate found in labs worldwide. If we include the monthly figure, this rises to 1 in 7 labs that regularly ignore their own QC flags. This is much higher than what is reported in the US (where the rate is about 1 in 28 labs). When QC is corrupted by poor practices, we're in danger of compromising patient results.
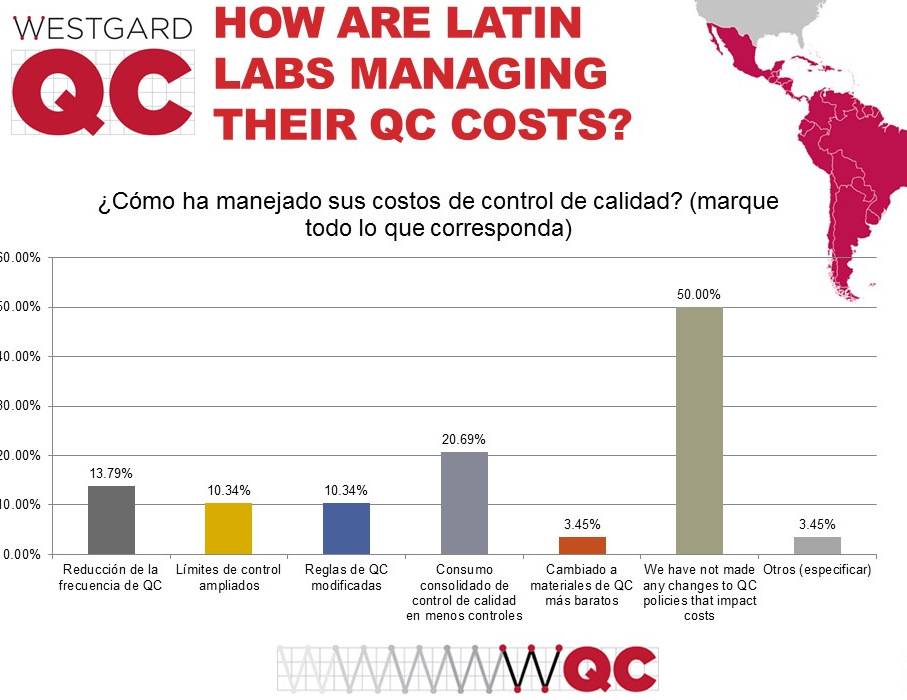
Finally, there is the question of resources and finances. Similar to the US and global labs, half of Latinn and South American laboratories have made no changes to their QC to manage costs. However distressing this is, it does mean there may be "low hanging fruit" for labs seeking cost savings and efficiency gains. Considering a change to consolidated or cheaper controls may be a way to reduce laboratory costs without compromising quality, when paired with the use of the right tools. And if the assays in the laboratory are of high quality, even more savings can be realized.
Conclusion
There are some encouraging findings in this survey, that practices may be a little bit better than the rest of the world. However, our colleagues at Quality Consulting (QSC) wanted us to take the survey with a grain of salt. There are many labs in Latin and South America, and only the better labs were likely to respond to the online survey. Labs where QC is not run regularly, where poor practices are in place, were less likely to respond. So we may only have a glimpse of how the best labs in the region are performing, not a true representation of all the laboratories. This is a flaw from which all surveys suffer. But even if we take this as a slice of the laboratories, it does mean that the top tier of this region's labs are running more "Westgard Rules" and trouble-shooting more appropriately, than labs in the rest of the world.
Overall, it's as sobering as the other regional and global surveys. There is major room for improvement in how QC is implemented and interpreted in Latin and South America. We hope that these results will spur laboratories to go beyond mere compliance, and strive for excellence, optimization, and true efficiency gains.
For a free copy of these graphs, go to our download section.