Sigma Metric Analysis
An Atellica HEMA 580 in South Korea
There's a new analyzer for hematology, the Atellica HEMA 580. A recent study looked at the its analytical performance in South Korea. Can the new analyzer meet the new CLIA 2025 goals?
An Atellica HEMA 580 in South Korea
May 2026
Sten Westgard, MS
[Sigma quality score: +0.5 for control levels at medical decision levels (abnormal, normal); +0.5 bias measured over long term period (796 patient samples); +0.5 actual patient samples used in bias estimation. Final score: 1.5 = Between Misleading and Optimistic.]
A recent study from Yonsei University Wonju College of Medicine in South Korea, examined the performance of the Atellica HEMA 580 hematology analyzer:
Analytical and Clinical Performance of the Atellica HEMA 580 Haematology Analyzer: A comparison with the ADVIA 2120i. Hyeongyu Lee, Kooyoung Cho, Gilsun Yoo, Juwon Kim. International Journal of Laboratory Hematology. 2026;0:1-11. https://doi.org/10.1111/ijlh.70107
Calculating Sigma metrics from routine laboratory performance data.
The imprecision and bias of the analyzers
"To evaluate reproducibility (between-run precision), three levels of commercial control materials (Level 1, 2 and 3) provided by the manufacturer were analyzer in duplicated for five consecutive days."
"The ADVIA 2120i and Atellica HEMA 580 results for the 796 whole blood specimens were compared in accordance with the CLSI EP09c guidelines.... Method comparison was performed using Passing-Bablok regression and Bland-Altman analysis."
In the study, RCPA QAP performance specifications were used. For our purposes, we will use CLIA 2025 goals, and for the analytes not directly regulated by CLIA, ESFEQA, and EFLM 2026. We also used the regression equation to calculate bias at each control level, rather than use a single bias average. Thus, our results will vary considerably with the study's results.
| HEMA 580 parameter | TEa Source | TEa | % Bias | CV | Sigma |
| Hemoglobin | CLIA 2025 4% | 4 | 5.70 | 1.17 | negative |
| 4 | 0.04 | 0.28 | 14.2 | ||
| 4 | 0.65 | 0.31 | 10.9 | ||
| Hematocrit | CLIA 2025 4% | 4 | 6.77 | 1.47 | negative |
| 4 | 0.85 | 0.75 | 4.2 | ||
| 4 | 1.81 | 0.87 | 2.5 | ||
| MCV | ESFEQA 10% | 10 | 0.38 | 0.57 | 16.8 |
| 10 | 0.98 | 0.44 | 20.3 | ||
| 10 | 1.45 | 0.71 | 12.1 | ||
| MCH | ESFEQA 9% | 9 | 1.12 | 0.71 | 11.2 |
| 9 | 0.99 | 0.53 | 15.2 | ||
| 9 | 0.94 | 0.57 | 14.2 | ||
| MCHC | ESFEQA 7% | 7 | 4.14 | 0.91 | 3.2 |
| 7 | 8.55 | 0.54 | negative | ||
| 7 | 7.53 | 0.79 | negative | ||
| Platelets | CLIA 2025 25% | 25 | 1.67 | 4.90 | 4.8 |
| 25 | 4.55 | 3.50 | 5.8 | ||
| 25 | 5.10 | 3.41 | 5.8 | ||
| RBC | CLIA 2025 4% | 4 | 0.90 | 1.28 | 2.4 |
| 4 | 1.97 | 8.72 | 0.2 | ||
| 4 | 2.06 | 10.05 | 0.2 | ||
| RDW | EFLM 2026 min 3.8% | 3.8 | 2.85 | 2.09 | 0.5 |
| 3.8 | 2.76 | 1.16 | 0.9 | ||
| 3.8 | 2.85 | 1.77 | 0.5 | ||
| Lymphocytes | EFLM 2026 min 22.4% | 22.4 | 4.34 | 4.39 | 4.1 |
| 22.4 | 4.08 | 2.86 | 6.4 | ||
| 22.4 | 4.89 | 4.86 | 3.6 | ||
| Monocytes | EFLM 2026 min 27.4% | 27.4 | 13.55 | 11.11 | 1.2 |
| 27.4 | 24.00 | 5.28 | 0.6 | ||
| 27.4 | 20.33 | 4.26 | 1.7 | ||
| Eosinophils |
EFLM 2026 min 44.6% | 44.6 | 2.87 | 6.71 | 6.2 |
| 44.6 | 2.07 | 4.61 | 9.2 | ||
| 44.6 | 2.16 | 4.03 | 10.5 | ||
| Basophils | EFLM 2026 min 27.3% | 27.3 | 19.61 | 1.41 | 5.5 |
| 27.3 | 19.54 | 0.86 | 9.0 | ||
| 27.3 | 19.56 | 0.87 | 8.9 | ||
| Neutrophils | EFLM 2026 min 26.3% | 26.3 | 2.20 | 1.99 | 12.1 |
| 26.3 | 2.13 | 1.36 | 17.8 | ||
| 26.3 | 1.67 | 0.86 | 28.6 | ||
| WBC | CLIA 2025 10% | 10 | 0.16 | 1.73 | 5.69 |
| 10 | 1.25 | 1.39 | 6.30 | ||
| 10 | 1.74 | 1.01 | 8.20 |
Graphic Display of performance by Normalized Method Decision Chart (NMEDx)
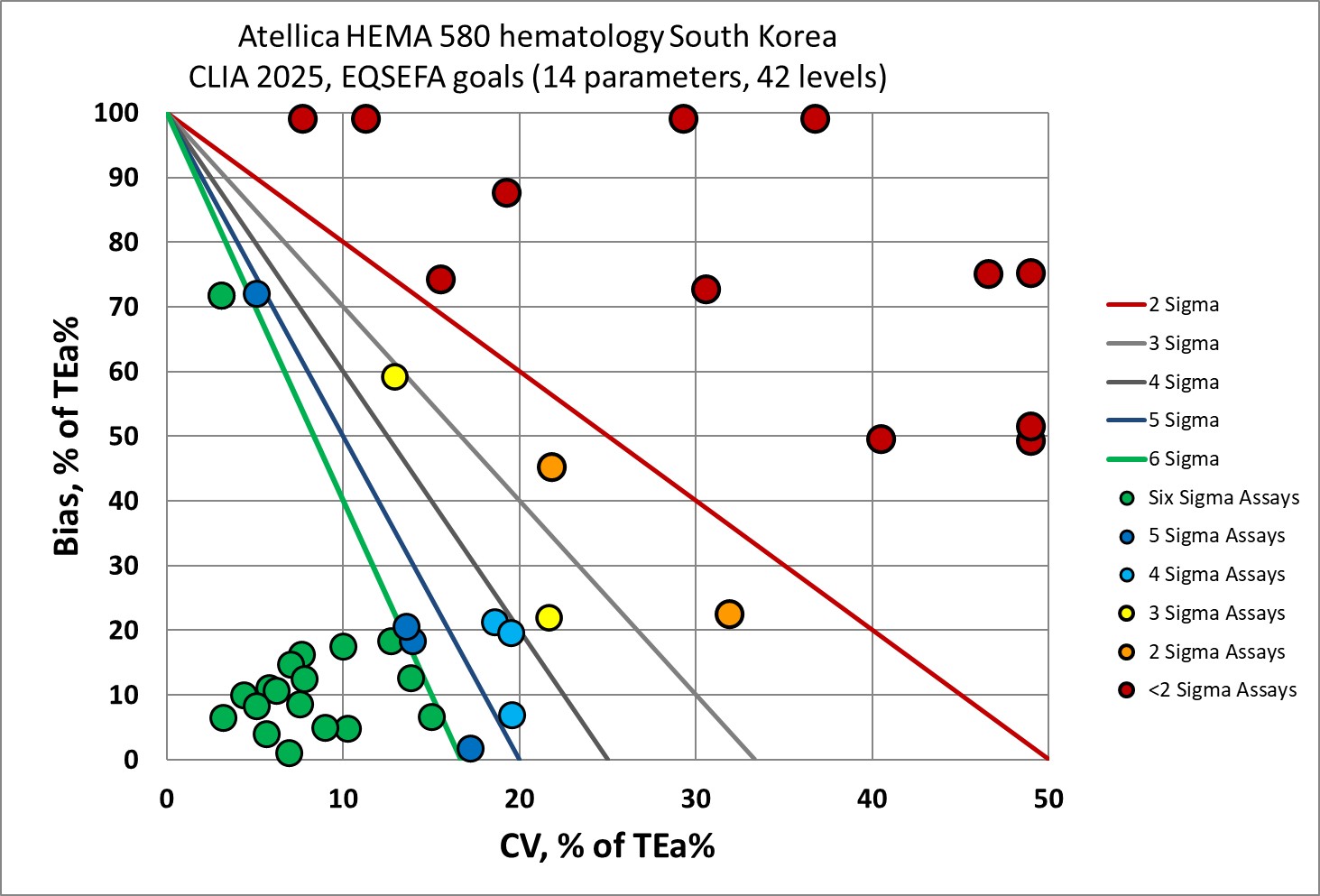
There's good news and bad news here. The low levels of hemoglobin and hematocrit are poor. The RBC, RDW and monocyte methods are uniformly poor. Overall, a third of the analytical performance of this instrument is below 3 Sigma.
However, there is a lot of Six Sigma performance here, too. The differential counts, which are typically quite poor in other studies, have great metrics in this study.
The additional limitation of this study is that the calculations are based on manufacturer controls, which makes them suspect. Third-party controls are strongly recommended. Hematology, of course, has fewer options to run such independent controls, and faces the additional challenge of short-term control life.
Conclusion
The authors of the study concluded: "The Atellica HEMA 580 demonstrated comparable analytical and clinical performance and strong correlation with the ADVIA 2120i. We conclude that the Atellica HEMA 580 is a competent HA [hematology analyzer] that can provide reliable and accurate diagnostic results in clinical laboratories."
Our conclusion would not go that far. Given the favorable context of using the ADVIA for comparison and using manufacturer controls, we would expect more optimistic metrics. The fact that even under these ideal circumstances a third of the performance is rated unacceptable (by CLIA and other global standards) means this instrument warrants caution and concern. A wise laboratory would seek out or demand additional performance data, collected under more rigorous conditions, before accepting it into routine use. We need to see more proof that it can deliver reliable results.