Quality of Laboratory Testing
Total Testing Process Sigma Metrics
Six Sigma metrics across the Total Testing Process in Thailand
May 2026
Sten Westgard, MS
A recent study examined 3 years of quality indicators across the Total Testing Process (TTP) at a general hospital in Thailand:
Analytical errors in the laboratory of a general hospital of the Thai Red Cross Society: A 3-year experience. Srivilai Trakulkaseamsiri and Kittipong Chumchujan. Journal of Associated Medical Sciences 2026; 59(1):33-39.
The authors reviewed three years of laboratory errors, from 2021 through 2023, encompassing 2,261,729 specimens.
For convenience, we summarize here the performance of all three phases of the TTP as Sigma metrics:
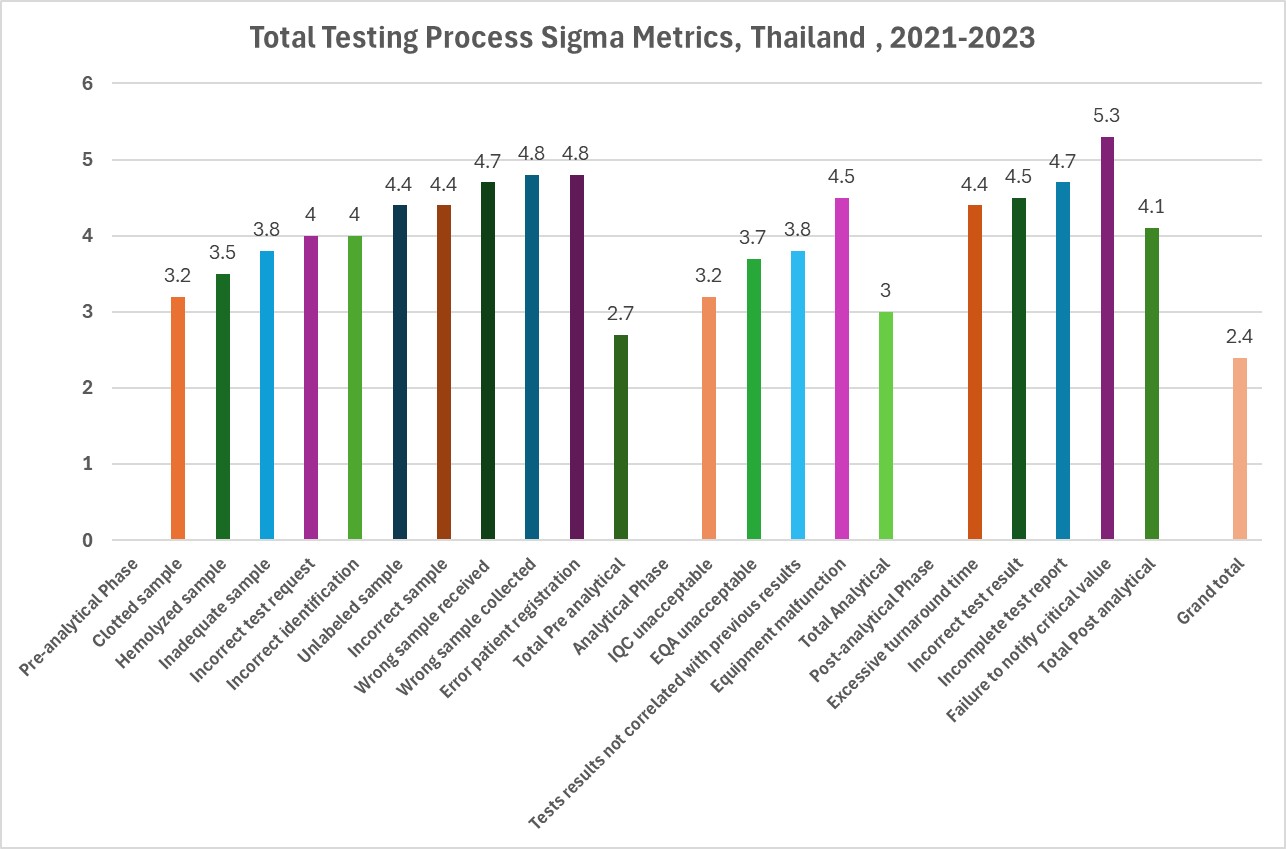
[click on the graphic to view it in a larger size]
The worst six processes are the following:
- Clotted sample (3.2 Sigma)
- IQC unacceptable (3.2 Sigma)
- Hemolyzed sample (3.5 Sigma)
- EQA unacceptable (3.7 Sigma)
- Inadequate sample (3.8 Sigma)
- Test results not correlated with previous results (3.8 Sigma)
Notice that half of the worst processes are from the analytical phase. IQC is essentially tied for the first/worst place.
If we summarize each phase of the TTP, these are the Sigma metrics:
- Pre-analytical phase: 2.7 Sigma
- Analytical phase: 3.0 Sigma
- Post-analytical phase: 4.1 Sigma
If you roll all of the errors together and grade the TTP as one unified process, it works out to a 2.4 Sigma. By conventional standards, therefore, the testing process is unacceptable in quality. But to be honest, if similar calculations were made for other laboratories, this metric would probably replicate across the globe. About 1 in 5 tests are impacted, in other words, by an error somewhere in the TTP. Approximately 1.4 errors occur in the pre-analytical phase every day, compared to 1 error in the analytical phase.
However, the way we count these errors matters, and distorts the impact and importance of different errors.
When a sample is clotted or hemolyzed, it is an individual specimen failure. Is that the case with IQC failure? or EQA failure?
No.
An IQC failure effects the entire analytical run. So one IQC failure might impact 10, 20, 30 specimens in that run. If we simply consider that minimal impact of 10 patient tests being impacted by an IQC failure, the number of defects per million increases from about 48,000 per million to 480,000 per million. That drops the Sigma metric down to 1.6.
If we take into account the way that analytical errors manifest themselves, they are the leading cause of errors, the lowest Sigma metric, and the worst part of the TTP.
Of course, that metric assumes that the IQC has been properly designed and implemented. If the typical IQC approach is used, a lot of those "failures" are actually false rejections. In complete absence of true analytical failures, the false rejection rate of 1:2s with 2 controls (which is 9%) would render the IQC process to be 2.9 Sigma. The fact that the study here pegs IQC failure at 3.2 may mean that the IQC is better than usual.
Another assumption is that the analytical methods are fit for purpose. Typical studies for TTP do not include an analytical Sigma assessment. They assume instead that all methods are fit for purpose. But if some of the methods being run are low in Sigma, or are not fit for purpose, they are generating hundreds, thousands, or even higher numbers of bad results. That's a blind spot of most TTP studies.
Pre-analytical quality matters. But so does analytical quality. A focus on improvements to IQC could yield a significant return on effort, making it one of the top priorities a laboratory should consider.