Guest Essay
Dietmar Stockl, QC Reality Check, Part Four
Part three of an ongoing series on Internal Quality Control (IQC) by Dietmar Stockl. Dr. Stockl looks again at QC data examples from the real world of the laboratory, this time looking at the case when a laboratory purposes uses the wrong standard deviation.
Internal Quality Control (IQC) – A reality check
Part IV: Purposely working with the “wrong standard deviation”
- Part I: Setting the scene
- Part II: “Pearls on a rope”
- Part III: Purposely working with the “wrong target”
- Part IV: Purposely working with the “wrong standard deviation”
- Part V: How variable/stable do I want it?
- Part VI: How stable can I get it?
- Part VII: What’s going on? – 1
DIETMAR STOCKL, PHD
MAY 2011
ReasoningWe have several immunoassays that regularly have instabilities that have no clinicical consequences. Therefore, we use the mid-term standard deviation for calculating control limits.
There is significant lot-to-lot variation of several immunoassys which, however, is also not considered clinically relevant. Lot durations are relatively short (1 to 3 months), therefore, we consider the establishment of lot-specific SDs and target values not as cost-effective. We calculate control limits with standard deviations that account for lot variations.
Lot-example
Short-term CV (1 lot) = 1.5%: 3s limits, short broken line
Mid-term CV (several lots): 3.1%: 2.58s limits, long broken, line.
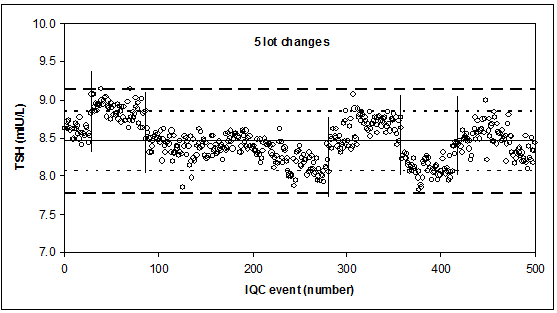
CAVE
Stable SD and target value (see Part III) are the heart of QC. If compromises are made at these places, one should be aware of the consequences thereof.
Do not forget: what you see in your IQC, typically, is reflected in your patient data. If IQC differs, your patients differ! Lot-to-lot variations will affect your patient data!
QC Problem with purposely (or instinctively) “wrong SD”
IQC results of an inflammatory protein (µg/mL), measured with a manual ELISA assay.
Problem description
“We are violating the 10X rule - which CLIA is concerned about”. “The violation might be because of the same vial being used over time”.
The chart
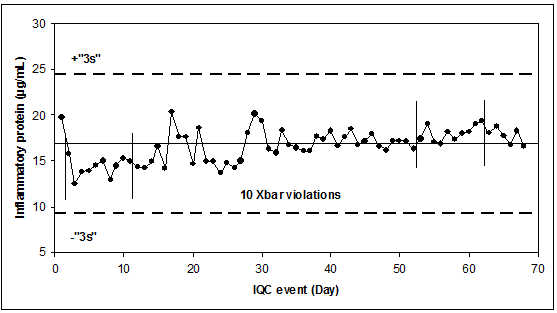
The data
The target value 16.83 was established before under stable conditions. The standard deviation of 2.53 was chosen to accommodate “typical instabilities” and lot-to-lot variations; however, the SD of a stable period is 0.9 (day 33 to 68), only!
IQC is done with a 3s rule, and additionally with a 10Xbar rule. There are 2 violations of the 10Xbar rule: in the beginning and in the end.
Reason for the violation
The reason is that the “stable” SD (SDstable) is much smaller than the SD used for the definition of the control limits (SDrule). Consequently, even medium-sized shifts/drifts will lead to violations of all rules that work with a mean (average rules) or with a location relative to the target (e.g., the 10Xbar-rule).
The solution
If SDstable << SDrule, average rules and X-bar rules are not a good choice for controlling the process.
Note: One should have a good justification for these wide limits; in principle, the QC limits are 8.4s limits when SDstable is used for calculation.